Technology Overview as part of a Web-based
Technical and Regulatory Guidance
Microbial Mats
1. Introduction
Click
Here to view case study table at the end of this document.
![]()
A microbial mat aquatic bioremediation system uses a naturally occurring,
living organism to rapidly remove metals from mining-impacted water (MIW).
Two very important aspects of microbial mats are their rapid growth rate
and ability to survive harsh environmental conditions such as high salinity
and low pH. Microbial mats also tolerate high concentrations of toxic compounds
that will kill plants or algae.
Although photosynthetic, the mats are not plants and are classified as bacteria based on their cellular structure. However, they are photosynthetic and can be grown like plants, harvested, and dried until needed. Constructed microbial mats are naturally occurring, living organisms composed primarily of cyanobacteria (formerly known as blue-green algae). Mats are nontoxic and do not produce offensive odors (Figure 1-1). They are called “constructed” mats because they are grown using a standard technique that is inexpensive and can be accomplished with minimal training.
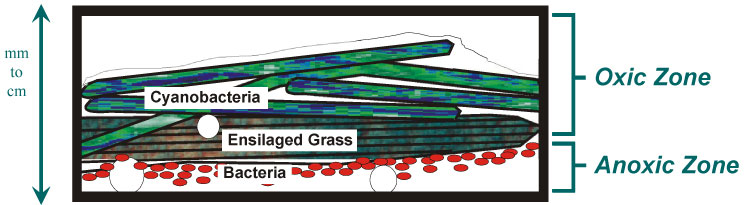
Figure 1-1. Cross section showing oxic and anoxic zone of microbial mats.
Photosynthetic microbial mats have few growth requirements (atmospheric carbon dioxide and water to produce oxygen and trace metals; they are nitrogen-fixing, i.e., use atmospheric nitrogen to make usable nitrogen compounds for their growth), and flourish even under harsh environmental conditions. The mats are photoautotrophs that can saturate the aqueous phase with oxygen (15 mg/L) during the daylight hours and have the potential to degrade organics through oxidative pathways (Figure 1-2). At night the mats consume the oxygen and provide an environment where organic contaminants can be reductively degraded (Nzengung et al. 2003; O’Niell and Nzengung 2000; O’Niell, Nzengung, and Adesida 2000; O’Niell et al. 2000; O’Niell et al. 1998; Murray, Phillips, and Bender 1997; O’Niell et al. 1997). Microbial mats have been shown to sorb dissolved phase organic contaminants from water in a matter of seconds to minutes (O’Niell, Nzengung, and Adesida 2000; O’Niell et al. 2000). Microbial mat treatment of water also results in low biological oxygen demand (BOD) and low dissolved organic carbon (DOC) levels in the water being treated.
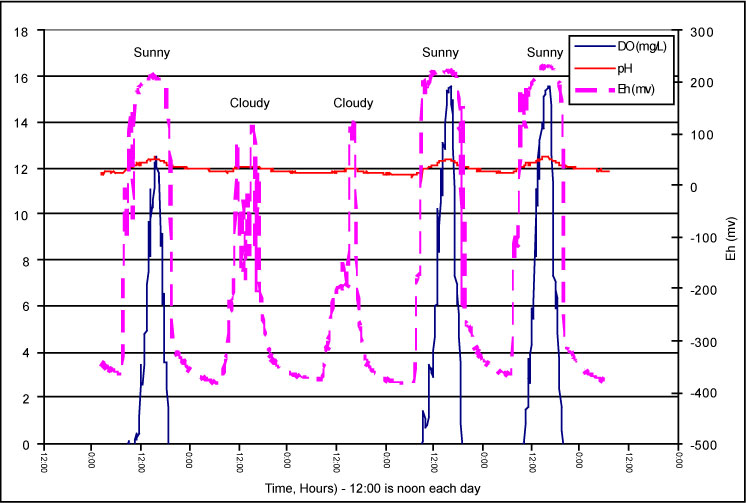
Figure 1-2. Natural diurnal variation in dissolved oxygen and oxygen-reduction potential levels in a microbial mat bioreactor.
Microbial mats also sorb and sequester a variety of metals, metaloids, radionuclides, and oxyanions from water through several different mechanisms:
- Ion exchange/sorption. Microbial mats provide a large
negatively charged surface area for binding positively charged metals.
Surface binding and ion exchange mechanisms are involved in the metal
removal process. Once all the binding sites are filled with metals, a
period of regrowth is needed to rejuvenate the microbial mats.
- Reduction. Many metals (e.g., U6+, Cr6+, and Se6+)
must be reduced for removal to occur. Metals are reduced in the anoxic
reducing zones created in microbial mats at night or in those maintained
in dark or low-light conditions. Once these metals are reduced, they
precipitate as oxides, hydroxides, or sulfides.
- Oxidation. Some metals (e.g., manganese) are oxidized
and precipitated. Microbial mats are photosynthetic and can saturate
the water with oxygen during daylight hours.
- Bioflocculants. Microbial mats produce negatively charged carbohydrate molecules that act as bioflocculants to bind metals and cause them to precipitate out of the water column.
2. Applicability
Can treat dissolved constituents (organics and inorganics).
- Can treat for a variety of metals, metaloids, radionuclides, and oxyanions.
- Can treat mine discharges collected in ponds and slow-flowing leachate.
- Can be used as a stand-alone technology or in combination with others.
Microbial mats can be applied for remediation purposes using several different techniques, and the costs of the natural materials used in the microbial mat-based system can be very cost-effective. Microbial mats can be grown on ensilaged (process of preserving green food for livestock in an undried condition in airtight conditions) grass in an aqueous solution as free-floating microbial communities. Alternatively, the microbial mats can be grown on the surface of a porous material such as cocoa fiber or glass wool. Another very promising technique is to immobilize the microbial mat on inert silica beads in a gelatinous form. Aqueous effluent contaminated with metals can then be mixed with the microbial mat gel. The metals bind to the negatively buoyant glass gel as it settles out of the mixture, and the contaminant-free water is decanted or pumped from the reaction vessel. As observed during pilot-scale test at the Savannah River Site (DOE/SR/18553-1), use of this technique caused 88% of uranium at an initial concentration of 2.4 mg/L to be removed from approximately 200 L of contaminated water in 15 minutes (Bender et al. 2000).
Microbial mats can also be grown, dried, and ground to powder for easy transport and application. Disposal costs can be minimized because a contaminated microbial mat can be harvested from bioreactors and dried to approximately 1%–2% of its wet mass, resulting in lower transport costs and less space required for landfills. Alternatively, the dried mat or mat/silica matrix can be combusted to ash, which is more easily disposed. It is also likely that the vitreous mat/silica bead matrix can be used as a fill or aggregate after air drying.
More details on the types of applications are provided in the following sections. All tests have been conducted at pilot scale or smaller.
2.1 Smaller-Scale
Applications
Microbial mats can be applied according to the scale of the problem. Potential
applications include the following:
- “Mat bags.” A dried microbial mat can be packaged in
bags designed like “tea bags.” The bags would be added to a container and
allowed to hydrate and sequester metals dissolved in the water. After a
prescribed period, the bag would be removed from the treated water.
- Low-cost filtration system. A multiplayer
filter bed can be constructed in a 200 L (55 gal) barrel. The lower third
of the drum contains clean filter sand to remove bacteria and suspended
particles, the middle third contains activated carbon (or alternatively
charcoal) to remove dissolved organic matter, and the top third contains
the microbial mat to remove metals. Parallel microbial mat filtration systems
can be maintained to ensure continuous operation, should one system require
maintenance.
- Silica beaded microbial mat reactor. A
microbial mat can be grown on a silica bead that is mixed with water and
buffered to make a microbial mat gel. Twenty liters of the beaded microbial
mat is then placed in a 200 L barrel. The barrel is filled with contaminated
water and stirred to mix with the beaded microbial mat. The negatively
buoyant gel is allowed to settle for 15 minutes, and the clean water is
decanted from the barrel. At the Savannah River Site in Aiken, SC, this
technique has been tested on water containing 2.4 mg/L uranium, and it
removed 88% of the uranium in 15 minutes. This system requires little maintenance
after setup. It can be maintained in an outdoor pond for two years with
no additional maintenance. The cost of raw materials for the silica gel
matrix is about US$3/L when purchasing a single ton, but the costs can
be reduced when purchasing larger volumes.
- Solar-powered “plate and shelf” bioreactor. At Fort Hood in Texas this type of bioreactor has been developed and field-tested for the removal of organic contaminants and metals (Figure 2-1). It consists of a 2m × 1m × 1m Lexan® box with five removable shelves and was built for approximately $3,000. The microbial mat bioreactor is operated at a rate of 2 gpm. The cyanobacteria are grown on fiber mats that fit onto each of the shelves. A pump is used to transfer the contaminated water into the top of the reactor. The water flows by gravity through each of the layers of microbial mat, the contaminants are removed, and clean water flows through an outlet at the base. This type of system can run for many years with minimal weekly maintenance.
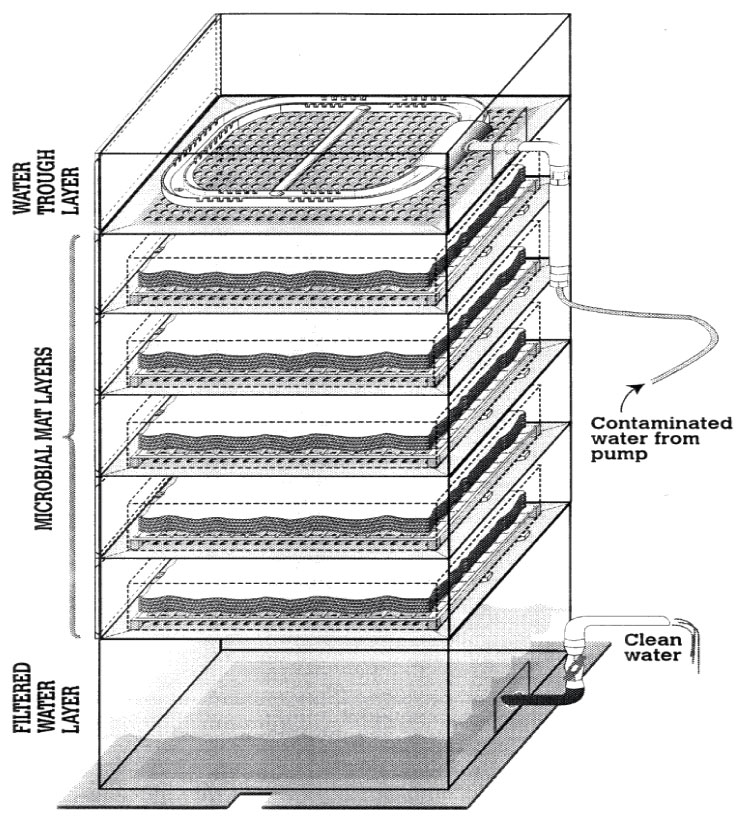
Figure 2-1. Schematic view of flow-through microbial mat plate and shelf bioreactor.
2.2 Larger-Scale
Applications
Microbial mats could be applied to larger water treatment systems using
various configurations. Microbial mats immobilized on silica beads can be
used in a series of large, stand-alone treatment cells or as a component
of a more conventional municipal water treatment plant. A microbial mat-based
system might operate as a large-scale fluidized-bed reactor to bind, precipitate,
and sequester metals. Alternatively, the microbial mat can be used as the
primary metal-removal component in a constructed wetland at a facility using
a total ecosystem–based treatment approach. PLANTECO has also field-tested
smaller biotreatment systems such as the solar-powered bioreactor and a low-cost,
low-tech bioremediation trench.
2.3 Full-Scale Microbial
Mat
An appropriate option for treatment of MIW is a combination microbial mat/constructed
treatment wetland system. At full scale, a passive microbial mat treatment
system would be designed to treat the MIW, which would then be polished with
a relatively small aerobic constructed treatment wetland (Fort Hood, TX case
study). A lime pretreatment should be incorporated in the design to raise
the pH and prevent iron precipitates from overwhelming the microbial mats.
Electric pumps may be required to transfer liquids from one phase of the
treatment process to another if the transfer cannot be achieved using gravity
flow. Should the MIW contain sufficient total organic carbon (TOC) and biodegradable
organic carbon, the addition of nutrients to the system will be minimized.
The microbial mats will feed off the organics and electron sources in the
leachate and continue to grow. Otherwise, the system may require supplemental
nutrient application. The growing microbial mat continuously increases the
surface area available for sequestering dissolved metals in the effluent
and increases the removal efficiency. Therefore, if a healthy microbial mat
is maintained, the role of the aerobic constructed treatment wetland (CTW)
will be mainly for effluent water quality polishing. A CTW can be constructed
using selected aquatic plants designed to remove the metals of concern at
the site. The components of the full scale system will include the following:
- chemical pretreatment to raise pH and remove excess iron
- modular microbial mat bioreactors in series to sequester targeted metals and biodegrade organic contaminants
- containerized or open constructed treatment wetlands for polishing
2.4 Conceptual Full-Scale Microbial
Mat Treatment System Design
A healthy microbial mat ecology is required
for the microbial mats to effectively remove the targeted metals from MIW
by sequestration, sorption, and precipitation. During the treatment process,
the microbial mat should continue to grow and produce fresh biomass. The
results of pilot field tests have shown that microbial mats can be impaired
by the deposition of precipitated metals (e.g., iron) and other suspended
solids on the photosynthesizing upper layer of the constructed microbial
mat (see Fort Hood, TX case study). A healthy microbial mat will increase
the dissolved oxygen content of the water to greater than 8 mg/L during
the day and will supersaturate the water with oxygen on sunny days (Figure
1-2). The dissolved oxygen increases the pH and oxidation-reduction potential
of the water to provide optimum geochemical conditions for precipitation
of iron, manganese and other redox-sensitive dissolved metals (Figure 1-2).
In the sulfate-reducing zones at the bottom of the floating mats, the metals
will precipitate as metal sulfides, especially during the night and winter
months when there is little or no photosynthesis. Also, if nutrient levels
in the effluent are lacking or minimal, additional nutrients should be
added to the system as part of a routine maintenance plan to promote microbial
mat growth and maximum efficiency. At night, reducing conditions predominate
if an artificial light source is not provided. Under reducing conditions
the performance of the microbial bioreactor is not expected to be as effective
as during daylight hours when the mats are actively photosynthesizing.
3. Advantages
- temporary or permanent applications
- low cost
- passive technology
- low energy and maintenance requirements
- potential for metal recovery
- decreases mass of waste disposal
The microbial mat bioremediation system provides several unique advantages over conventional techniques:
- Low cost. Microbial mats can be grown
at very low costs. Ensilaged grass and silica bead are both very inexpensive
components.
- Solar driven. These systems are controlled by the amount
a sunlight available, thus having fewer power requirements to implement
then other technologies.
- Minimal training required. The application of the technology
does not require any special skills.
- Low maintenance. A simplified treatment system can be
designed using either minimal or no moving parts.
- Ease of application. Technology is easily transportable
to other locations in a country.
- Applicable to harsh environmental conditions. Mats can
survive in cold temperatures and will flourish in temperatures above 25°C
with abundant sunlight. They can be dried and will reform upon rehydration.
Microbial mats are unaffected by high concentrations of many contaminants.
Unlike many plants or algae, microbial mats can survive high, often phytotoxic
concentrations of metals or organic contaminants.
- Reduction of waste byproducts and sludge. Because microbial mats can be dried to 1%–2% of their wet volume, they will not produce as much waste as coagulation/precipitation processes, adsorption processes, or ion exchange processes. It should be possible to recycle the vitric crust of the dried mat/silica gel–based treatment system to reduce disposal costs or the need to landfill the by-product.
4. Limitations
- Chemical
characteristics of the influent mine water:
- generally not effective for low pH (pH < 2)
- generally not effective for high concentrations of Fe, Mn, Al
- may require pretreatment to remove Fe
- Suspended solids in the wastewater need to be removed prior to treatment.
- Trace metal toxicity may inhibit the microbial viability of some constructed
.microbial mats. This could be determined in treatability/optimization
tests.
- Hot climates with abundant sunshine or artificial light source make for
optimum microbial mat growth and optimum system performance.
- Only pilot-scale data are available; full-scale application for some of the system designs described is lacking.
5. Performance
Metals that have been successfully removed from mine-influenced water streams
by microbial mats include Ag, As, Ba, Cd, Co, Cr, Cu, Fe, Mn, Ni, Pb, Sr,
and Zn (see TVA, Alabama case study), including acid mine drainage (Bender
et al. 2000; Bender and Phillips 1995; Bender et al. 1994a, 1994b; Phillips
et al. 1993; Bender et al. 1991; Bender and Ibeanusi 1987). Research indicates
that microbial mats can also effectively remove dissolved uranium and mercury
from other types of wastewaters (Bender et al. 2000; O’Niell and Noakes,
unpublished). Extensive research has proven that metals are rapidly removed
from solution by microbial mats. Microbial mats provide large negatively
charged surface areas for sequestration of metals and also provide highly
oxic photosynthesizing zones in close proximity to anoxic reducing zones
where metals can be precipitated. Positively charged metals (e.g., lead [Pb2+])
are attracted to the negatively charged surfaces of the microbial mat, where
surface binding and ion exchange mechanisms prevent their release. Other
metals (U6+, Cr6+, and Se6+) are reduced in the anaerobic zones of microbial
mats and are precipitated from the solution eventually bound in the microbial
mat itself. Bender et al. (1994a, 1994b) found that manganese was oxidized,
precipitated, and sequestered by microbial mats (see TVA, Alabama case study).
Microbial mats also produce negatively charged bioflocculants which rapidly
bind metals and remove them from the water column through flocculation.
6.
Costs
Although there are currently limited data available to calculate the life-cycle
cost of microbial mat aquatic treatment systems, the per gallon cost for
most acid mine waste (AMW) ranges from $0.35 to $0.90 per 1000 gallons. The
life-cycle cost of a microbial mat aquatic treatment system would significantly
decrease if pretreatment is used to remove suspended matter ahead of the
microbial mat treatment. Reduction in cost (i.e., operation and maintenance)
may be achieved by mining the spent microbial mat for economic metals, using
the biomass for bioenergy production, and using solar or wind energy to meet
the energy requirements at the treatment facility.
7. Regulatory Considerations
Metals in AMW are incorporated into the microbial mats and become hyperaccumulated.
Thus, at most sites the spent microbial mat may be classified as a hazardous
waste under the Resource Conservation and Recovery Act. Once dried, a microbial
mat is composed of 3% solids. Disposal of the reduced mass of spent dried
microbial mat as a hazardous or nonhazardous waste should be addressed up-front
with local regulatory agencies. If metals are recovered from the spent microbial
mats, the residual biomass can be used for soil enhancement or bioenergy
production or disposed as a nonhazardous waste at a landfill.
8. Stakeholder Considerations
Depending on the type and amount of metal removed, the spent microbial mat
may be processed in multiple ways for economic benefits. The spent microbial
mat may be processed to recover some of the sequestered metals that are of
economic value. The microbial mats biomass may also be fermented to produce
biogas used to generate heat at the treatment facility during winter months.
9. Lessons Learned
Although constructed microbial mats are capable of adapting to sudden changes
in composition of the aqueous media (pH and chemical composition) by reconfiguring
its different components (i.e., microbial composition), deposition of precipitated
metals or sludge on the microbial mat’s upper surface causes loss of viability
and function (see Fort Hood case study). An effective design of a microbial
mat treatment system should ensure that the precipitated metals accumulate
in the anaerobic sulfate-reducing bottom layer or are actively incorporated
into the mat from solution rather than by surface deposition.
For very high concentrations of metals exceeding aqueous solubility, pretreatment to precipitate out metals tends to increase the useful life of the microbial mat (lifetime in pounds removed) and reduce the change-out time. Some metals may be precipitated out of the solution and accumulate in the treatment pond rather than incorporated into the microbial mat. To address the latter scenario, a recovery system should be provided to recover the precipitated metals.
For some AMW solutions with low concentrations of dissolved organic carbon, supplying the microbial mats with a cheap source of natural organic carbon should sustain the microbial mat growth and treatment effectiveness. This organic carbon could be ensilaged grass or a cheap nutrient rich solution.
Sunlight intensity is an important requirement, and contingencies should be provided for artificial light sources when sunlight is not available for extended periods.
Like all biological systems, system performance decreases during winter seasons. Locating the microbial mat treatment system in a greenhouse should ensure predictable high performance year round.
During field operations the rate of microbial mat usage may be higher than predicted, so a readily available stock of constructed microbial mats should be maintained for such eventualities.Table 10-1. Case studies using microbial mats
11.
References
Bender, J., M. C. Duff, P. Phillips, and M. Hill. 2000. “Bioremediation
and Bioreduction of Dissolved U(VI) by Microbial Mat Consortium Supported
on Silica Gel Particles,” Environmental Science and Technology 34(15):
3235–41.
Bender, J., J. P. Gould, Y. Vatcharapijarn, and G. Saha. 1991. “Uptake, Transformation and Fixation of Se(VI) by a Mixed Selenium Tolerant Ecosystem,” Water, Air, and Soil Pollution 59(3–4): 359–67.
Bender, J., J. P. Gould, Y. Vatcharapijarn, S. Young, and P. Phillips. 1994b. “Removal of Zinc and Manganese from Contaminated Water with Cyanobacterial Mats,” Water Environment Research 66(5): 679–83.
Bender, J., and V. Ibeanusi. 1987. “Effects of Supplements on the Bioaccumulation of Lead in Anabaena Spp.,” Bulletin of Environmental Contamination and Toxicology 39(2): 209–13.
Bender, J., R. F. Lee, and P. Phillips. 1995. “A Review of the Uptake and Transformation of Metals and Metalloids by Microbial Mats and Their Use in Bioremediation,” Journal of Industrial Microbiology 14(2): 113–18.
Bender, J., and P. Phillips. 1995. “Biological Remediation of Mixed Waste by Microbial Mats,” Federal Facilities Environmental Journal 6(3): 77–85.
Bender, J., J. R. Washington, B. Graves, P. Phillips, and G. Abotsi. 1994a. “Deposit of Zinc and Manganese in an Aqueous Environment Mediated by Microbial Mats,” Water, Air, and Soil Pollution 75(3–4): 195–204.
Murray, R., P. Phillips, and J. Bender. 1997. “Degradation of Pesticides Applied to St. Vincent Banana Farm Soils Comparing Native Bacteria and Microbial Mat,” Environmental Toxicology and Chemistry 16(1): 84–90.
Nzengung. V. A., W. L. O’Niell, N. L. Wolfe, D. E. Rennels, and S. C. McCutcheon. 2003. “Removal of Halogenated Organic Compounds Dissolved in Water by Algae, Aquatic Plants and Microbial Mats,” in Phytoremediation: Transformation and Control of Contaminants, S. C. McCutcheon and J. S. Schnoor, eds. Wiley-Interscience.
O’Niell, W. L., and V. A. Nzengung. 2000. “Treatment of Organic Contaminated Water Microbial Mat Bioreactors,” pp. 247–52 in Symposium Series: Case Studies in the Remediation of Chlorinated and Recalcitrant Compounds 2(4), G. B. Wickramanayake, A. R. Gavaskar, J. T. Gibbs, and J. L. Means, eds. Columbus, OH: Battelle Press.
O’Niell, W. L., V. A. Nzengung, and A. Adesida. 2000. “Treatment of Perchlorate Contaminated Water in Microbial Mat, Algae, and Ebb and Flow Hydroponic Bioreactors,” pp. 101–106 in Symposium Series: Case Studies in the Remediation of Chlorinated and Recalcitrant Compounds 2(7), G. B. Wickramanayake, A. R. Gavaskar, J. T. Gibbs, and J. L. Means, eds. Columbus, OH: Battelle Press.
O’Niell, W. L., V. A. Nzengung, J. E. Noakes, J. Bender, and P. Phillips. 2000. “Biosorption and Transformation of Tetrachloroethylene and Trichloroethylene Using Mixed Species Microbial Mats,” Journal of Hazardous Substance Research2(2): 1–16. www.engg.ksu.edu/HSRC/JHSR/vol2no2.pdf
O’Niell, W. L., V. A. Nzengung, J. E. Noakes, J. Bender, and P. Phillips. 1998. “Biodegradation of Tetrachloroethylene and Trichloroethylene Using Mixed Species Microbial Mats,” pp. 233–37 in Proceedings, 1st International Conference on Remediation of Chlorinated and Recalcitrant Compounds, May 18–21, Monterey, CA.
O’Niell, W. L., V. A. Nzengung, J. E. Noakes, J. Bender, and P. Phillips. 1997. “Biodegradation of Tetrachloroethylene and Trichloroethylene Using Mixed Species Microbial Mats,” Proceedings, Conference on Emerging Technologies in Hazardous Waste Management IX, September 15–17, Pittsburgh.
Phillips, P., J. Bender, R. Simms, S. Rodriguez-Eaton, and C. Britt. 1993. “Manganese Removal from Acid Coal-Mine Drainage by a Pond Containing Green Algae and Microbial Mat,” Proceedings, 2nd International Symposium on Waste Stabilization Ponds and Reuse of Pond Effluents, Water Science Technology 31(12): 161–70.
U.S. Environmental Protection Agency. 2000. Constructed Wetlands Treatment of Municipal Wastewaters. EPA/625/R-99/010.
