Technology Overview as part of a Web-based
Technical and Regulatory Guidance
Electrokinetics
1. Introduction
Click
Here to view case study table at the end of this document.
![]()
The electrokinetic remediation (ER)
process removes metals and organic contaminants from low-permeability soil,
mud, sludge, and marine dredging. ER uses electrochemical and electrokinetic
processes to desorb, and then remove, metals and polar organics. This in
situ soil-processing technology is primarily a separation and removal technique
for extracting contaminants from soils (Figure 1-1).
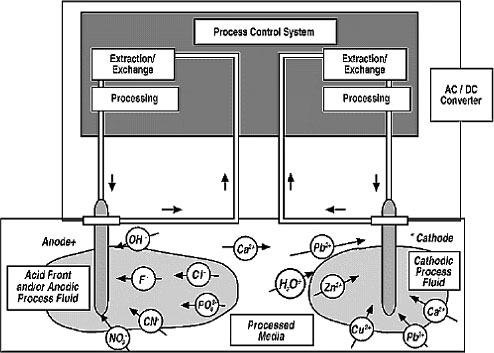
Figure 1-1. Dimensions of an electrokinetic system.
2. Applicability
Targeted contaminants for electrokinetics
are heavy metals, anions, and polar organics in soil, mud, sludge, and
marine dredging. Contaminant concentrations that can be treated range from
a few parts per million (ppm) to tens of thousands ppm.
3. Advantages
Some advantages are as follows:
- can remove dissolved and sorbed contaminants from low permeability matrix
- applied in situ with little surface disturbance
- wide range of contaminant concentration can be treated
4. Limitations
Factors that may limit the applicability and effectiveness of this process
are as follows:
- Effectiveness is sharply reduced for wastes with a moisture content of less than 10%. Optimal effectiveness occurs if the moisture content is between 14% and 18%.
- The presence of buried metallic or insulating material can induce variability in the electrical conductivity of the soil; therefore, the natural geologic spatial variability should be delineated. Additionally, deposits that exhibit very high electrical conductivity, such as ore deposits, cause the technique to be inefficient.
- Inert electrodes, such as carbon, graphite, or platinum, must be used so that no residue will be introduced into the treated soil mass. Metallic electrodes may dissolve as a result of electrolysis and introduce corrosive products into the soil mass.
- Electrokinetics is most effective in clays because of the negative surface charge of clay particles. However, the surface charge of the clay is altered by both charges in the pH of the pore fluid and the adsorption of contaminants. Extreme pH at the electrodes and oxidation-reduction changes, induced by the process electrode reactions, many inhibit ER’s effectiveness, although acidic conditions (i.e., low pH) may help to remove metals.
- Oxidation/reduction reactions can form undesirable products (e.g., chlorine gas).
5. Performance
There have been few, if any, commercial applications of electrokinetic
remediation in the United States. The electrokinetic technology has been
operated for test and demonstration purposes at the pilot scale and at
full scale at the following sites:
- Louisiana State University
- Electrokinetics, Inc.
- Geokinetics International, Inc.
- Battelle Memorial Institute. Geokinetics International, Inc. has successfully demonstrated the in situ electrokinetic remediation process in five field sites in Europe.
In 1996, a comprehensive demonstration study of lead extraction at a U.S. Army firing range in Louisiana was conducted by the Department of Defense’s Small Business Innovative Research Program and Electrokinetics, Inc. EPA, taking part in independent assessments of the results, found pilot-scale studies have demonstrated that concentrations of lead decreased to less than 300 mg/kg in 30 weeks of electrokinetic processing when the soils where originally contaminated as high as 4500 mg/kg of lead.
6.
Costs
Costs vary with the amount of soil to be treated, the conductivity of
the soil, the type of contaminant, the spacing of electrodes, and the type
of process design employed. Ongoing pilot-scale studies using “real-world”
soils indicate that the energy expenditures in extraction of metals from
soils may be 500 kWh/m3 or more at electrode spacing of 1.0–1.5 m. Direct
costs estimates of about $15/m3 for a suggested energy expenditure of $0.03
per kilowatt hours, together with the cost of enhancement, could result
in direct costs of $50/m3 or more. A recent study estimated full-scale
costs at $117/m3.
7.
Regulatory Considerations
None reported.
8. Stakeholders Considerations
None reported.
9. Lessons Learned
None reported.
Table 10-1. Case studies including electrokinetics treatment
Case Study Name |
Case Study Location |
| Electrokinetic Remediation at Alameda Point, Alameda, CA | http://costperformance.org/profile.cfm?ID=5&CaseID=5 |
| Electrokinetic Extraction at the Unlined Chromic Acid Pit, Sandia National Laboratories, NM | http://costperformance.org/profile.cfm?ID=246&CaseID=246 |
| Electrokinetics at Site 5, Naval Air Weapons Station Point Mugu, CA | http://costperformance.org/profile.cfm?ID=189&CaseID=189 |
| In Situ Electrokinetics Remediation at the Naval Air Weapons Station, Point Mugu, CA | http://costperformance.org/profile.cfm?ID=188&CaseID=188 |
11. References
U.S. Army Environmental Command. 1997. “In Situ Electrokinetic
Remediation for Metal Contaminated Soils,” pp. 87–88 in Innovative
Technology Demonstration, Evaluation and Transfer Activities, FY 96 Annual
Report. SFIM-AEC-ET-CR-97013.
U.S. Department of Energy. 1995. “Electrokinetic
Remediation of Heavy Metals and Radionuclides,” pp. 201–203 in Technology
Catalogue, 2nd ed. DOE/EM-0235.
Office of Environmental Management, Office of Technology Development.
USEPA (U.S. Environmental Protection Agency). 1997. Recent Developments
for In Situ Treatment of Metal Contaminated Soils.
www.clu-in.org/download/remed/metals2.pdf.
USEPA. 1998. Guide to Documenting and Managing Cost and Performance Information for Remediation Projects. EPA-542-B-98-007.
USEPA. 2000a. Abstracts of Remediation Case Studies, vol. 4. EPA-542-R-00-006. www.epa.gov/tio/download/frtr/abstractsvol4.pdf.
USEPA. 2000b. Innovative Remediation Technologies: Field-Scale Demonstration Projects in North America, 2nd ed. EPA-542-B-00-004. Office of Solid Waste and Emergency Response. www.clu-in.org/download/remed/nairt_2000.pdf.
USEPA. 2001. Treatment Technologies for Site Cleanup: Annual
Status Report, 10th ed. EPA-542-R-01-004. Office of Solid Waste and
Emergency Response.
